Breezula: Überblick, Studienergebnisse und FDA-Zulassungsdatum
Trotz des raschen Fortschritts in den meisten medizinischen Bereichen ist das Gebiet der androgenetischen Alopezie in den gesamten 2000er Jahren in einen Dornröschenschlaf verfallen. Seit 1997 hat die FDA kein neues Medikament zur Behandlung dieser Krankheit zugelassen, die geschätzte Allein in den USA sind 50 Millionen Männer und 30 Millionen Frauen betroffen.
Zum Glück für die Betroffenen gibt es ein neues Medikament am Horizont, das geduldig auf seine FDA-Zulassung wartet.
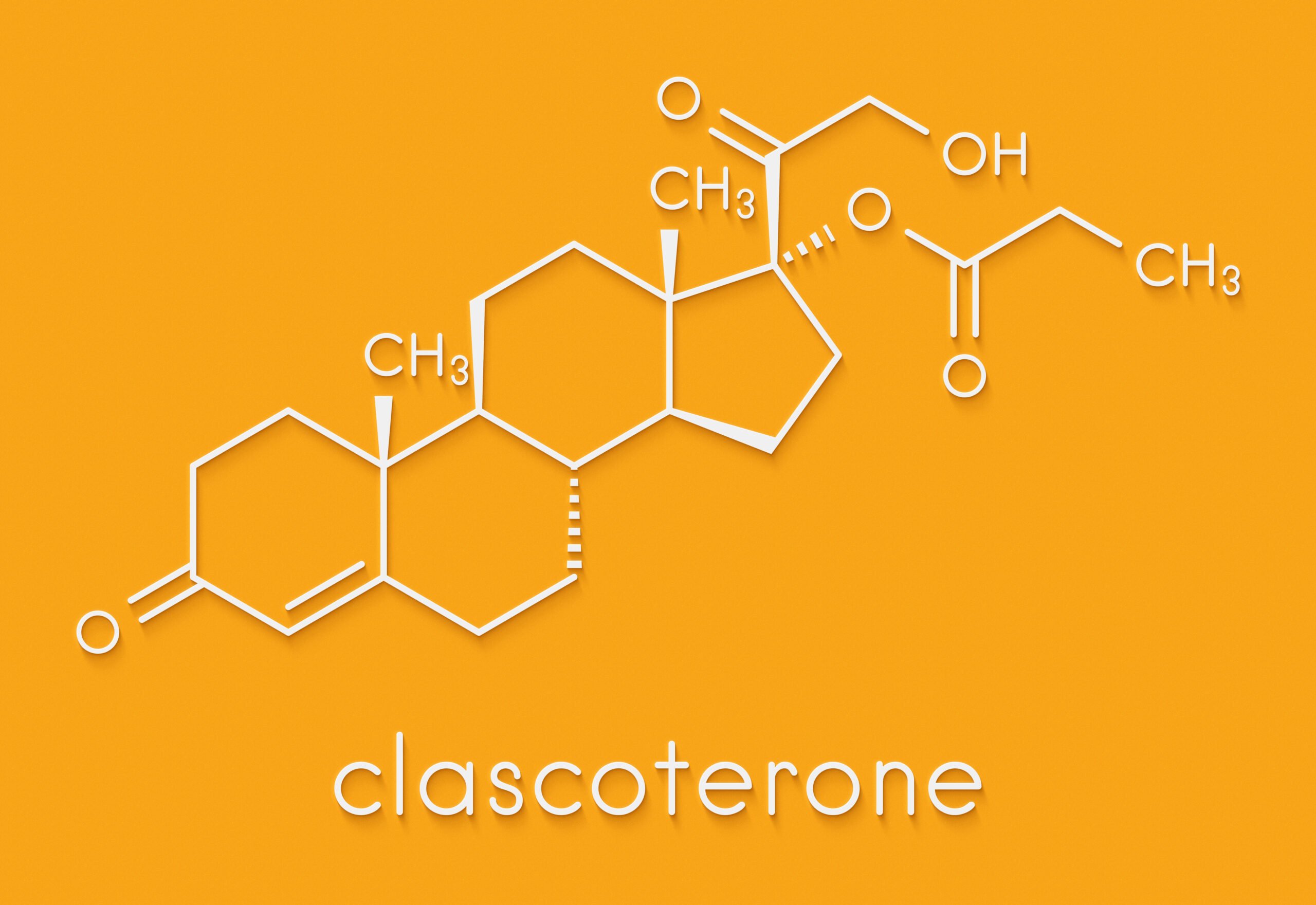
Im Jahr 2019 kündigte das italienische Pharmaunternehmen Cassiopea ein neues Medikament an, das vermutlich den Haarausfall stoppt und Haare nachwachsen lässt. Sie kündigten das Medikament unter dem Namen Clascoterone mit dem Markennamen Breezula an.
Berichten zufolge hemmt Breezula die Androgenrezeptoren, wodurch der Haarausfall gestoppt und das Wachstum von neuem Haar gefördert wird. Es befindet sich derzeit noch in der Testphase und ist nicht käuflich zu erwerben. Mal sehen, wie lange es noch dauert, bis es auf den Markt kommt!
Was ist Breezula?
Breezula ist der Markenname von Clascoterone, einem neuen Medikament, das von Cassiopea entwickelt wurde, einem italienischen Pharmaunternehmen, das sich auf medizinische Dermatologie spezialisiert hat.
Androgenetische Alopezie entsteht häufig durch eine übermäßige Reaktion des Körpers auf die Produktion von Androgenen, den Sexualhormonen, die einen großen Einfluss auf den Haarzyklus und die Geschwindigkeit des Haarwachstums haben.
Bei einer übermäßigen Reaktion auf diese Hormone werden die Haarfollikel überstimuliert, wodurch das Haar brüchig und schwach wird, was häufig zu Kahlheit und dünner werdendem Haar führt.
Breezula wirkt, indem es die Androgenrezeptoren in unserem Nervensystem blockiert und so Haarausfall verhindert. Es ist eine topische Behandlung, d. h. es wird direkt auf die Kopfhaut aufgetragen wie topisches Minoxidil.
Obwohl einige Formen des Medikaments für die Behandlung von Akne vulgaris (Winlevi) zugelassen sind, ist es von der FDA noch nicht als Mittel gegen Haarausfall zugelassen.
Das Medikament Clacosteron ist für die Behandlung von Akne in einer Konzentration von 1% zugelassen, aber für Haarausfall wird es in Konzentrationen von 2,5%, 5% und 7,5% getestet, so dass der Testprozess intensiv ist und mehr Zeit benötigt.
Wie funktioniert Breezula?
Der Grundgedanke von Breezula ist derselbe wie bei Finasterid - es blockiert die Androgene, die für den Haarausfall verantwortlich sind, aber das ist nur die einfachste Erklärung. In Wirklichkeit ist der Prozess komplizierter als das, aber wir werden ihn nach und nach erläutern.
Der Hauptverursacher: DHT
Die Hauptverantwortlichen für Haarausfall und Glatzenbildung sind Androgene, eine Familie männlicher Hormone, die für das Haarwachstum verantwortlich sind. Das einflussreichste Hormon aus dieser Familie ist Dihydrotestosteron oder DHT, wie es allgemein bekannt ist.
DHT ist der Hauptgrund für zahllose haarbezogene Probleme, einschließlich androgenetischer Alopezie, Haarausfall und Haarverdünnung. Es bindet sich an die Androgenrezeptoren in den Haarfollikeln und verursacht zahlreiche biochemische Reaktionen, die zum Schrumpfen dieser Follikel führen.
Geschrumpfte oder kranke Haarfollikel führen zu dünner werdendem Haar und schließlich zu Haarausfall bei Männern und Frauen.
Deshalb haben die meisten Haarwuchsmittel das gleiche Ziel: die DHT-Rezeptoren zu blockieren, um zu verhindern, dass sich das Hormon überhaupt erst bindet, und so den Haarausfall rückgängig zu machen.
Der Breezula-Effekt
Wenn Sie Breezula direkt auf Ihre Kopfhaut auftragen, wird es schnell metabolisiert und bindet sich an die Androgenrezeptoren in den Haarfollikeln, da es in seiner Form dem DHT ähnelt. Es aktiviert die Rezeptoren jedoch nicht, so dass die Haarfollikel unbeeinflusst bleiben.
Gleichzeitig kann sich das DHT nicht binden, weil das Medikament die Rezeptoren blockiert, so dass der Haarausfall aufhört.
Wie unterscheidet sich Breezula von Finasterid?
Breezula und Finasterid wirken beide auf die gleiche Weise, indem sie die DHT-Wirkung blockieren. Wie unterscheidet sich Breezula also von der bereits etablierten oralen Behandlung, die seit mehr als 25 Jahren von der FDA zugelassen ist?
Finasterid blockiert zwar die DHT-Wirkung, aber auf andere Weise. Anstatt sich an die Androgenrezeptoren zu binden, zielt es auf die Bildung von DHT ab. Es verhindert, dass Testosteron in DHT umgewandelt wird, was zu einer Verringerung des DHT-Spiegels in Ihrem Körper führt.
Finasterid ist wissenschaftlich wirksamer als Breezula, weil es bereits alle möglichen klinischen Studien durchlaufen hat und zugelassen wurde. Es garantiert ein positives Ergebnis, was man von Breezula nicht sagen kann, bevor es nicht getestet wurde.
Trotzdem kann Breezula gegenüber Finasterid einen Vorteil haben, weil es nicht die gleichen Nebenwirkungen verursacht. Finasterid ist dafür berüchtigt, dass es schnelle Stimmungsschwankungen und sexuelle Funktionsstörung. Als topische Behandlung hat Breezula keine Auswirkungen auf den Sexualtrieb. Es kann zu Nebenwirkungen kommen, die aber wahrscheinlich mit der Haut zusammenhängen, wie Rötungen, Juckreiz usw.
Wie verlaufen die Breezula-Tests?
Breezula wird bereits seit mehr als vier Jahren getestet. Derzeit wird es sowohl in den USA als auch in Deutschland getestet, und die Ergebnisse der Phase-III-Studie sollen 2024 bekannt gegeben werden.
Solange die offiziellen Ergebnisse nicht vorliegen, kann das Medikament noch nicht zugelassen werden. Wir werden also sehen, wie der Testprozess verläuft und wann er voraussichtlich abgeschlossen sein wird:
Die Phase-II-Studie
Im Jahr 2019 veröffentlichte Cassiopea die Ergebnisse einer 12-monatige Probezeit die an Männern durchgeführt wurde, die an androgenetischer Alopezie leiden. Der Test umfasste drei Konzentrationen von Breezula: 2,5%, 5% und 7,5%.
An der Studie nahmen 404 Personen im Alter zwischen 18 und 55 Jahren mit unterschiedlich starkem Haarausfall teil. Einige Männer wendeten das Medikament ein ganzes Jahr lang auf die kahlen Stellen ihrer Kopfhaut an, während andere das Placebo anwendeten.
Die Ergebnisse zeigten positive Ergebnisse in Bezug auf die Verhinderung von Haarausfall und das Wachstum neuer Haare. Auch in Bezug auf lokale Hautreaktionen und unerwünschte Wirkungen schnitt es gut ab.
Die Phase-III-Studie
Auf der Grundlage der Ergebnisse der Phase II-Studie wählte Cassipea ursprünglich die 7,5%-Konzentration von Breezula für das Phase-III-Studiendie für Juni 2023 angekündigt waren. Später wurde jedoch beschlossen, sie zu ändern und stattdessen 5% Breezula in größeren Mengen zu verwenden.
Die Studien sollten eigentlich schon früher durchgeführt werden, wurden aber durch die Pandemie verzögert. Sie bestehen aus einer 6-monatigen multizentrischen, prospektiven, randomisierten, doppelbindenden, Vehikel-kontrollierten Studie, gefolgt von einer einfach bindenden Behandlung für weitere 6 Monate.
Diese Studie wird mit insgesamt 1500 Männern wesentlich umfangreicher sein als die erste, aber solange die Ergebnisse nicht bekannt sind, kann das Medikament nicht für den Markt zugelassen werden.
Wann wird Breezula von der FDA zugelassen?
Das Datum der FDA-Zulassung ist noch nicht bekannt, weder von Cassiopea noch von der FDA selbst. Dazu muss die klinische Prüfung von Breezula erst mit der Veröffentlichung der offiziellen Ergebnisse abgeschlossen werden.
Danach wird das Überprüfungsteam der FDA die Daten analysieren, sie gründlich prüfen und zu einer Entscheidung darüber gelangen, ob der Nutzen die Risiken überwiegt.
Wenn die Phase-III-Studie, wie vom Unternehmen beabsichtigt, im Juni 2024 abgeschlossen wird, kann es bis zur FDA-Zulassung noch etwa sechs Monate dauern, mehr oder weniger. Das bedeutet, dass das Medikament hoffentlich gegen Ende 2024 oder Anfang 2025 zugelassen wird.
Einige Arzneimittel werden in anderen Ländern zugelassen, aber nicht in den USA, und einige Arzneimittel werden für andere Zwecke als die Behandlung von Haarausfall zugelassen und dann off-label eingesetzt. Breezula könnte sich in einer ähnlichen Situation befinden, je nachdem, wie die verbleibenden Studien ausfallen.
Was sind die Nebenwirkungen von Breezula?
Da die Versuche mit Breezula noch nicht abgeschlossen sind, sind die Nebenwirkungen noch nicht bekannt. Nach Abschluss der Phase-II-Studie gab Cassiopea bekannt, dass es keine unerwünschten Wirkungen gab, die in direktem Zusammenhang mit dem Behandlungsmedikament stehen. Mit anderen Worten: Das Medikament ist sicher und nicht mit riskanten Nebenwirkungen verbunden.
Unter eine Studie für 2020 des Medikaments Clascoterone zeigte sich, dass die Nebenwirkungen gering und ungefährlich waren, aber es handelte sich um das Medikament zur Bekämpfung von Akne und nicht um die Version gegen Haarausfall.
Solange das Medikament nicht von der FDA für den Markt zugelassen ist, werden wir die Nebenwirkungen nicht mit Sicherheit kennen, da keine offizielle Liste veröffentlicht wird.
Es ist jedoch wahrscheinlich, dass Breezula aufgrund seiner Wirkungsweise keine schweren Nebenwirkungen haben wird. Wenn Clascoteron in unserem Körper verstoffwechselt wird, produziert es Cortexolonein Metabolit, der natürlich im menschlichen Körper vorkommt und keine Risiken birgt. Darüber hinaus hat Cassiopea festgestellt, dass Breezula keine Auswirkungen auf das Cortisol hat, wie es bei einigen Medikamenten der Fall ist, und daher keine Bedenken aufkommen lässt.
In einer Studie wurde ein ähnliches Cortexolon-Präparat untersucht, und als mögliche Nebenwirkungen wurden Akne, Überempfindlichkeit und Rötungen festgestellt. Das bedeutet aber noch nicht, dass Breezula die gleichen Nebenwirkungen verursacht.
Breezula im Vergleich zu anderen topischen Behandlungen
Derzeit sind die einzigen von der FDA für die Behandlung von Haarausfall zugelassenen Medikamente topisches Minoxidil und Finasterid. Dutasterid wird jedoch auch als Off-Label-Behandlung eingesetzt, was bedeutet, dass es für eine andere Erkrankung verschrieben, aber zur Behandlung von Haarausfall verwendet wird. Sehen wir uns an, wie diese Behandlungen funktionieren und wie Breezula im Vergleich dazu wirkt:
Finasterid
Finasterid ist häufig die erste Wahl bei der Behandlung von Haarausfall, insbesondere bei Haarausfall bei Männern. Es verhindert weiteren Haarausfall, weil es die DHT-Konzentration in Ihrem Körper reduziert, indem es das für die DHT-Synthese verantwortliche Enzym 5AR blockiert.
Wie Minoxidil ist Finasterid sowohl in topischer als auch in oraler Form erhältlich, und für beide ist ein ärztliches Rezept erforderlich. Außerdem wird es nur Männern verschrieben, da es bei Frauen zahlreiche Nebenwirkungen hervorruft, wie z. B. unregelmäßige Menstruation, Brustschwellungen und vermehrte Körperbehaarung. Auch bei männlichen Anwendern kann es zu Nebenwirkungen kommen, z. B. Erektionsstörungen und Kopfschmerzen, die jedoch weniger schwerwiegend sind.
Finasterid ist wahrscheinlich die ähnlichste Behandlung Droge zu Breezule aus dieser Liste. Beide haben einen ähnlichen Mechanismus zum Stoppen des Haarausfalls.
Minoxidil
Minoxidil ist sowohl in oraler als auch in topischer Form erhältlich, und es hat sich bei der Behandlung von Haarausfall als sehr wirksam erwiesen. Topisches Minoxidil ist rezeptfrei erhältlich, während die orale Form wie Finasterid verschreibungspflichtig ist. Allerdings muss es off-label sein, da orales Minoxidil nicht von der FDA für die Behandlung von Haarausfall zugelassen ist.
Obwohl Minoxidil ständig mit Finasterid verglichen wird, funktioniert es auf eine völlig andere Art und Weise, d. h. es unterscheidet sich auch von der Wirkungsweise von Breezula.
Minoxidil zielt auf die Blutgefäße ab und erweitert sie, um den Blutfluss zu den Haarfollikeln zu erhöhen, wodurch diese stimuliert und zum Haarwachstum angeregt werden.
Seine topische Form hat einige leichte Nebenwirkungen wie Trockenheit, Kopfhautreizung und Schuppenbildung. Unterdessen kann orales Minoxidil Schwindel, geschwollene Augen, Kopfschmerzen und Schlaflosigkeit verursachen, aber es hängt von jedem Patienten und seiner Krankengeschichte ab.
Dutasterid
Dutasterid hat die gleiches Behandlungskonzept wie Finasterid. Es wirkt ähnlich, indem es das Enzym namens 5-Alpha-Reduktase blockiert, das für die Produktion von DHT verantwortlich ist. Allerdings zielt es nicht wie Finasterid auf das Isoenzym vom Typ 2 ab, sondern sowohl auf Typ 1 als auch auf Typ 2 5AR.
Dutasterid hat eine stärkere Wirkung als Finasterid und wird daher häufig in schweren Fällen eingesetzt, in denen Finasterid keine großen Fortschritte erzielt. Außerdem zielt es auf die beiden Isoenzyme ab und ist daher effizienter bei der Umkehrung der Auswirkungen von geschrumpften Haarfollikeln.
Im Vergleich dazu kann Dutasterid mehr als 90% des DHT-Syntheseprozesses blockieren, während Finasterid nur 70% blockiert. Das Medikament kann sowohl oral eingenommen als auch topisch angewendet werden.
Wo steht Breezula?
Wenn wir die Behandlungen gegen Haarausfall von der höchsten bis zur niedrigsten Effektivität einstufen, würden wir sagen, dass Dutasterid am effektivsten ist, gefolgt von Finasterid und Minoxidil an letzter Stelle. Obwohl Breezula noch nicht ausreichend getestet wurde, gehen wir davon aus, dass es bei der Behandlung von Haarausfall genauso wirksam ist wie Finasterid, da es auf die gleiche Weise wirkt.
Minoxidil mag an letzter Stelle stehen, wenn es darum geht, Haarausfall zu verhindern, aber es ist die wirksamste Option, wenn es darum geht, gesünderes Haar wachsen zu lassen. Durch die Stimulierung der Haarfollikel regt es das Wachstum neuer Haare an, die schneller und dicker sind als zuvor.
Wir glauben nicht, dass Breezula die gleiche Wirkung in Bezug auf das Nachwachsen der Haare hat, aber das müssen wir noch sehen.
Abschließende Überlegungen
Obwohl sich Breezula noch in der Testphase befindet, gibt es bereits große Hoffnungen für das möglicherweise erste Mittel gegen Haarausfall, das seit zwei Jahrzehnten von der FDA zugelassen wird. Das Medikament ist derzeit nur in niedrigen Konzentrationen zur Behandlung von Akne erhältlich, aber es ist noch nicht in seiner Form zur Behandlung von Haarausfall verfügbar.
Bis die Phase-III-Studie abgeschlossen ist, werden wir nicht mit Sicherheit wissen, ob Breezula die FDA-Zulassung erhalten wird, aber wir sind gespannt auf die Möglichkeiten!